Sweet and sticky: Using Sugars to Develop Regenerative Therapies.
By LifETIME CDT Student: Amaziah Alipio (University of Birmingham)
Deaths from chronic and advanced liver disease such as liver cirrhosis and cancer accounts for over 2.4% of total deaths globally in 2017. The only effective therapy for chronic liver diseases is liver transplantation, however, the lack of available donor organs is a critical limitation in therapy. Regenerative medicine is a scientific field that utilises the use of stem cells for the functional restoration and regeneration of damaged tissues and organs. These cells are characterised by self-renewal and differentiation into specific cell types. Stem cell-based therapies are emerging as a more efficient, less invasive, and readily available treatment option compared to organ transplantation.
One of the significant hurdles scientists are facing is the lack of long term effectiveness of these cells. In most clinical trials, cell-based therapies are directly injected into target tissue and local vasculature, with the aim of achieving sufficient cell engraftment regenerate the damaged tissue. However, effectiveness can be limited by cell death caused by the shock of transferring cells from standard cell culture media to the damaging local microenvironment. Poor engraftment of injected cells to the desired location can also be attributed to the blood circulation resulting in cells being washed away from the site. Often, fewer than 5% of injected cells persist at the site of injection after a few days of transplantation.
Fig. 1 – Alginate is a polysaccharide which can be harvested from giant kelp Macrocystis pyrifera.
One approach that has been used to improve therapeutic cell engraftment is through the use of biomaterial carriers. Hydrogels are a class of biomaterials that are composed of long chain water-binding polymeric networks which can either be synthetic (e.g., poly(ethylene glycol) or PEG) or naturally derived such as the seaweed polysaccharide, alginate. Polysaccharides are large molecules composed of many smaller monosaccharide sugars. Hydrogels can be engineered to undergo gelation during or immediately after injection or undergo a “shear-thinning” process during injection. Simply put, hydrogels can act as a system to increase the viscosity of injected cell solutions and act to position cells in the desired site for attachment or act as a shield to the potentially toxic microenvironment of diseased tissues.
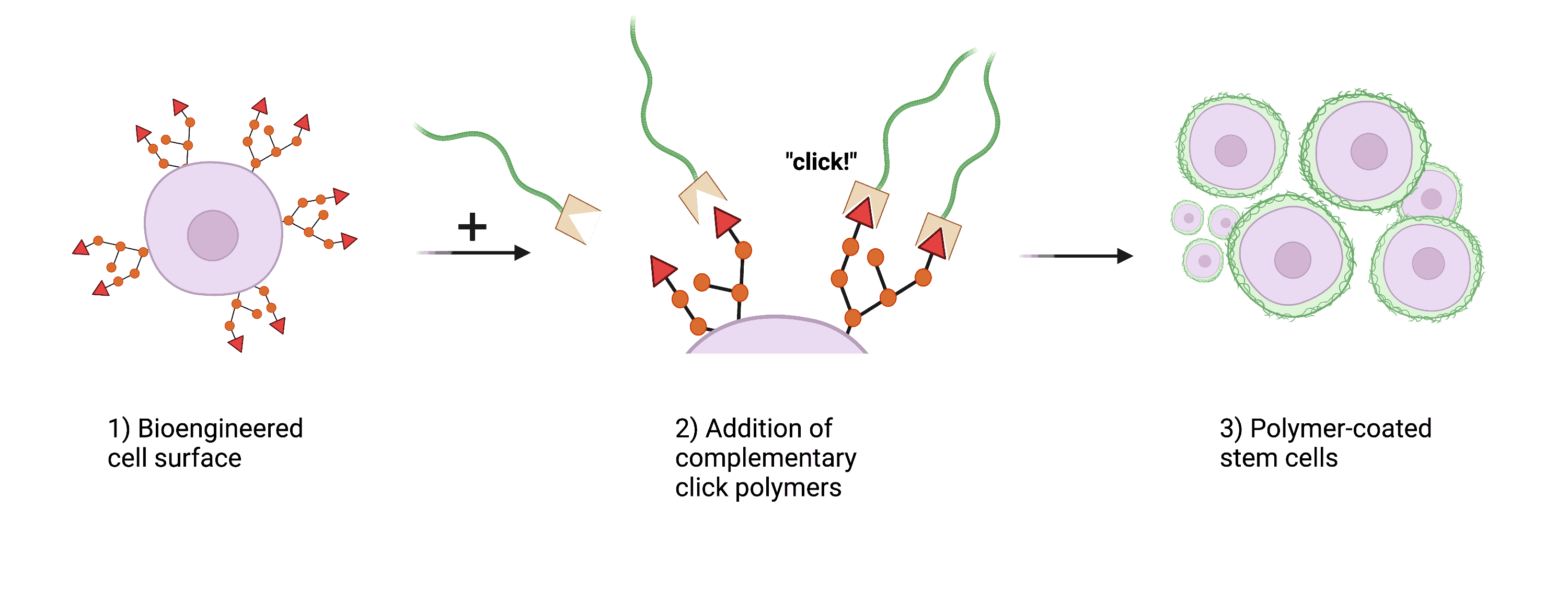
Fig. 2 – A schematic of our approach to achieve artificial cell coatings.
My project aims to explore the potential of polymeric biomaterials as cell coatings to increase stem cell viability and improve binding to host tissue. We plan to do this by engineering cells to display unnatural surface groups at the cell surface, following this, we can then “click” modified polysaccharide and other polymers onto the surface to create an artificial coating to modulate cell behaviour. The “click” reactions used in our approach are as fast as the name suggests. In addition to reaction rate, these click-reactions are biologically compatible and can allow for the control of polymer density and distribution. This work may have the potential to improve the therapeutic effectiveness and clinical use of cell-based therapies, offering a versatile platform for the manipulation of cell behaviour and inclusion of abiotic functionalities.