Your VISION is our MISSION
By LifETIME CDT Student: Seyedmohammad Moosavizadeh (University of Galway)
Defective corneal wound healing is a critical medical problem and an area of largely unmet medical need. Infections, persistent inflammation, trauma, and systemic disorders such as diabetes can all impair ocular wound closure and leave millions of people blind worldwide. Interestingly, the cornea is the most highly innervated tissue in the body and corneal injury and inflammation can lead to the local degeneration of corneal nerves which are vital for our vision.
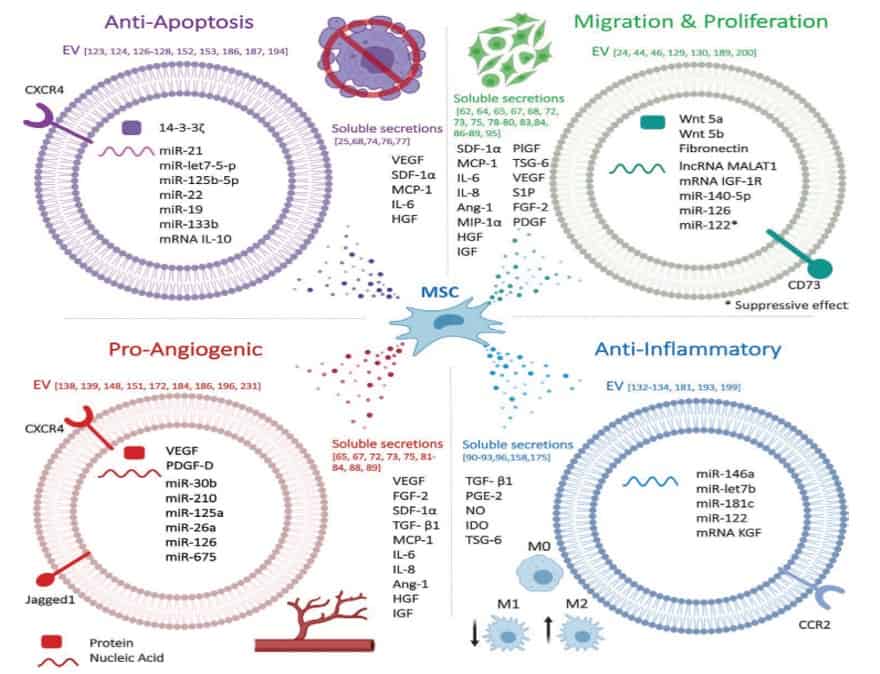
There are different treatments for ocular injuries such as anti-inflammatory drugs, surgeries, membrane grafting, and cell therapies. Corticosteroids are the mainstay for treatment of ocular inflammation and injuries; however, they are largely ineffective for the treatment of serious ulcers and neurodegeneration, and, in addition, have many unwanted side effects. Surgeries and membrane grafting are not patient friendly and have a range of side effects too. Mesenchymal stromal cells (MSC) have been shown to modulate inflammation and to facilitate ocular repair and to promote cell and nerve regeneration by upregulation of gene clusters encoding for neurotrophic factors. However, the risk of cell therapies for the treatment of severe diseases should not be under-estimated as large numbers of cells may have to be applied in order to obtain a therapeutic effect which might also enhance the potential risk for tumorigenesis. Extracellular vesicles (EVs) are smallparticles secreted from all cell types which are involved in intracellular communication. Depending on the cell of origin they contain various cargos. It has been demonstrated that EVs released from MSC (MSC-EVs) mediate some of the MSC therapeutic effects such as immunomodulation and regeneration. Researchers found that MSC-EVs promote corneal cell proliferation in-vitro and topical administration of MSC-EV enhances ocular wound healing. However, maintaining their biological activity and sustained and on-target release are the main obstacles in EV therapies.
Biomaterials are promising solutions to address the EVs therapy limitations by encapsulating the MSC-EVs and release them at the site of action in a controlled manner. Among the different types of biomaterials in which EV-infusion is exploited, hydrogels have proven to be the most user-friendly, economical, and accessible material. Treatment of ocular injuries and neurodegeneration with therapeutic MSC-EV embedded in a hydrogel for controlled release could therefore lead to a novel, cell-free treatment option for patients suffering from corneal injury and neurodegeneration.In this study, we aim to isolate extracellular vesicles (EVs) from human mesenchymal stromal cells (MSC) and investigate their therapeutic potential. Also, we wish to develop a biocompatible biomaterial which can encapsulate and deliver the MSC-EVs to the site of action with a controlled release profile. Finally, we aim to study the therapeutic and regenerative potentials of the functionalized biomaterial in in-vitro 2D and 3D corneal cell- and nerve injury models.
We hypothesise that this multidisciplinary project can provide a novel cell-free ocular injury treatment without unwanted side effects and with the highest therapeutic effects. The final product may have the potential clinical and commercial applications as well.








![Figure 2. Biomaterial functionalization and in-vitro therapeutic investigation [bioRENDER]](https://lifetime-cdt.org/wp-content/uploads/2022/11/Picture-2.jpg)